This study reports how Endocalyx Pro improves endothelial glycocalyx health, with improvement within the group each month during the four-month study. These scores were calculated by using GlycoCheck testing technology, a non-invasive test using a high-resolution video camera that tracks the movement of red blood cells and how they interact with the endothelial glycocalyx. In just minutes, GlycoCheck identifies and analyzes 3,000,000 blood vessel sites.
This test calculates capillary density, microvascular red blood cell content, and the glycocalyx barrier function to determine a person’s overall MicroVascular Health Score™.
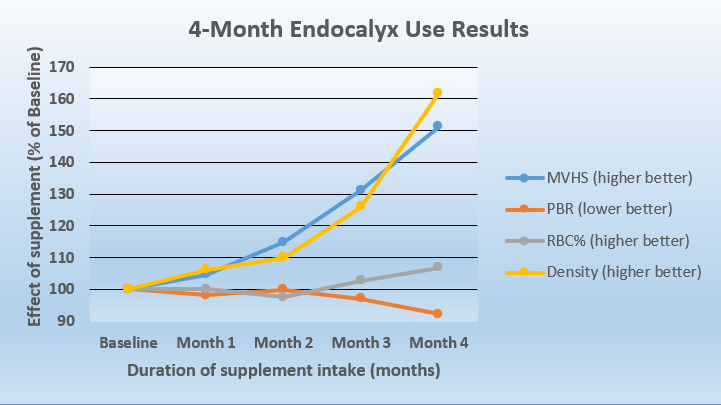
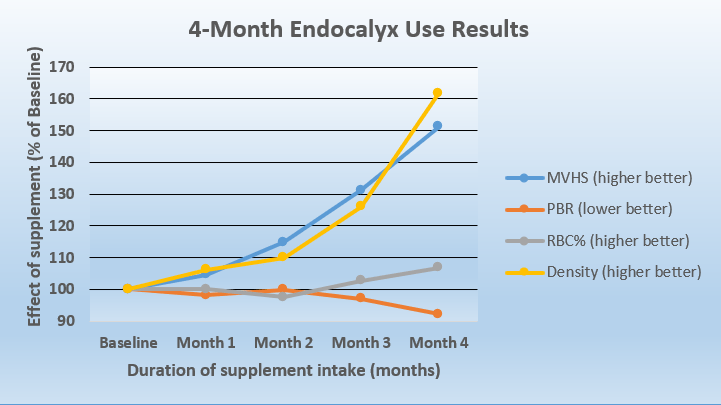
As indicated in the chart below, baseline microvascular values, before Endocalyx Pro was started are set at 100. The key data point is the overall “MVHS Score” which increased by 51.3%, to an index of 151 after four months. The test tracks:
- Capillary Density (Density): This is the number of blood vessels with more than 50% red blood cell content. A higher number is better.
- Red Blood Cell Filling Percent (RBC%): This is the average red blood cell content of individual blood vessels. A higher number is better.
- Perfused Boundary Region (PBR): Part of the glycocalyx that allows red blood cell access. A healthy glycocalyx protects the vessel wall and prevents circulating blood cells from getting too close to the vessel wall surface. When the glycocalyx is compromised, red blood cells penetrate deeper in the glycocalyx and blood vessels are more vulnerable. A LOWER number is better.
- Microvascular Health Score (MVHS): Overall score of microvascular health. A higher number is better.
As shown in this study and documented in the following chart, after one month of taking Endocalyx Pro daily, numbers improved. The most notable improvement began in month two, continuing through month four. The study was only planned for four months and demonstrates progressively improving scores over the whole period.
The study included both women and men, age 40 to 83. They included a cross-section of health issues including heart disease, diabetes (both type 1 and type 2), hypertension, and kidney disease. The goal was to have a typical cross section of American consumers that would be potential customers for this product.
They were asked not to change their diet during the test, and all provided a list of medications and health conditions. All participants continued to take the medications as prescribed by their doctor. Three participants were on blood thinners. They had their normal PT tests and the blood stayed within the desired thickness range.
Some participants reported that their blood pressure readings were reduced into better ranges. These individuals worked with their personal physician to decrease the hypertensive medication they were on.
There were no adverse effects reported by any of the individuals during the four-month study.